Unraveling the Latest Discoveries in DNA Repair
Unraveling the Latest Discoveries in DNA Repair
Exploring the Frontiers of DNA Repair: A Pioneering Journey
In the ever-evolving landscape of scientific discovery, the field of DNA repair stands at the forefront of groundbreaking revelations. This article delves into the latest strides in unraveling the intricacies of DNA repair mechanisms, shedding light on the profound implications for medicine, genetics, and beyond.
Understanding the Essence: What is DNA Repair?
DNA repair, a fundamental process within our cellular framework, plays a pivotal role in maintaining genomic stability. It acts as a guardian, tirelessly fixing damaged DNA strands, thereby safeguarding the integrity of our genetic code. Let’s embark on a journey to unravel the mysteries surrounding this intricate cellular mechanism.
The Symphony of Molecular Machines
DNA Helicases: Unraveling the Double Helix
At the core of DNA repair are molecular machines known as DNA helicases. These intricate enzymes perform the remarkable task of unwinding the double helix structure, allowing access to the damaged regions. Their precision and efficiency in this process are key to initiating the subsequent repair steps.
Polymerase Power: Stitching the Genetic Fabric
Another crucial player in the DNA repair orchestra is DNA polymerase. Think of it as the skilled tailor stitching up the torn fabric of our genetic code. This enzyme diligently synthesizes new DNA strands, filling in the gaps created by damaged sections. This meticulous repair process ensures the restoration of the original genetic blueprint.
Revolutionizing Medicine: Implications of DNA Repair Discoveries
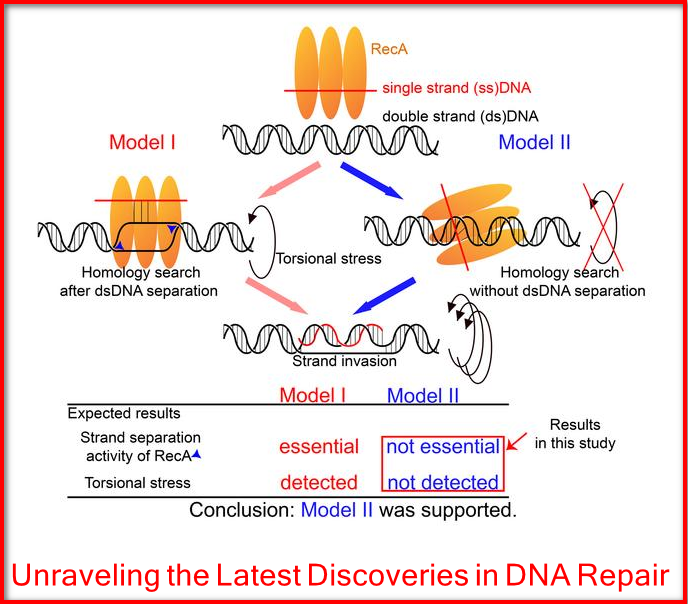
Researchers from Tokyo Metropolitan University have been studying DNA repair by homologous recombination, where the RecA protein repairs breaks in double-stranded DNA by incorporating a dangling single-strand end into intact double strands, and repairing the break based on the undamaged sequence.
They discovered that RecA finds where to put the single strand into the double helix without unwinding it by even a single turn.
Homologous recombination (HR) is a ubiquitous biochemical process shared across all living things, including animals, plants, fungi, and bacteria.
During HR, one of the two exposed ends of the break in the helix falls away, revealing an exposed single-stranded end; this is known as resection.
Then, a protein known as RecA (or some equivalent) binds to the exposed single strand and an intact double strand nearby.
Next, the protein “searches” for the same sequence. When it finds the right place, it recombines the single strand into the double helix in a process known as strand invasion, according to the study published in the journal Nucleic Acids Research.
The broken DNA strand is subsequently repaired using the existing DNA as a template.
A team led by Professor Kouji Hirota of Tokyo Metropolitan University sought to test two competing models for what happens when HR occurs.
In one, RecA unwinds a section of the double strand during the “homology search,” where it tries to find the right place for strand invasion to occur. In the second, there is no unwinding after the binding of RecA; only when strand invasion takes place does any unwinding occur.
The team, in cooperation with a team from the Tokyo Metropolitan Institute of Medical Science, adopted two approaches to tackle which of these actually happens.
Detailed insights into homologous recombination are vital to understanding what happens when things go wrong.
For example, factors implicated in breast cancer (BRCA1 and BRCA2) are also responsible for the correct loading of single-stranded DNA onto RAD51, the human version of RecA.
This suggests that problems with HR might underlie high incidences of breast cancer in patients with hereditary defects in BRCA1 or BRCA2.
The team hopes that findings like theirs will lead to new directions for research into cancer, the authors said
Source: AAAS
Dear Andrea
I would like to express my sincere gratitude for your insightful article. Your thoughtful analysis and well-researched content have provided me with valuable information and a deeper understanding of the topic. I appreciate the effort you put into crafting such a compelling piece.
Thank you once again for sharing your knowledge and expertise.
Best regards,
JOUHIN